We are an innovative and ambitious med-tech company that is working tirelessly to improve the quality of life of patients suffering from a variety of Vertebral Fractures

Our Story
Amber Implants is a VC-backed med-tech company started its operation in 2019.
The founders of the company, Dr. Banafsheh Sajadi and Dr. Mohammad Ahmadi have accomplished their Ph.D. degrees at Delft University of Technology and started developing innovative and creative orthopaedic implants since then. The company is now consisting of a (rapidly expanding) young and ambitious team that works on the developments of orthopaedic products. Also, a well-experienced and internationally-known team are already planning for commercialization of the product in the next stage. The company is working according to ISO 13485 and FDA guidelines for the manufacturing of the medical devices.
Our first product, VCFix® Spinal System has passed all pre-clinical validation and has started the clinical phase in 2023.
Our mission
Quality
At Amber Implants we are dedicated to deliver high quality medical products and services to improve patients’ quality of life.
Commitment
Amber Implants is committed to and accountable for advancing diversity, equity, and inclusion in all of its forms.
Team
At Amber Implants we prioritize to build and maintain a healthy and equally treated organization with highly qualified employees.
Our team
Company timeline

Approval received from both the Ethics Committee (EC) and BfArM in Germany for our pivotal EXPAND study of the VCFix® Spinal System.
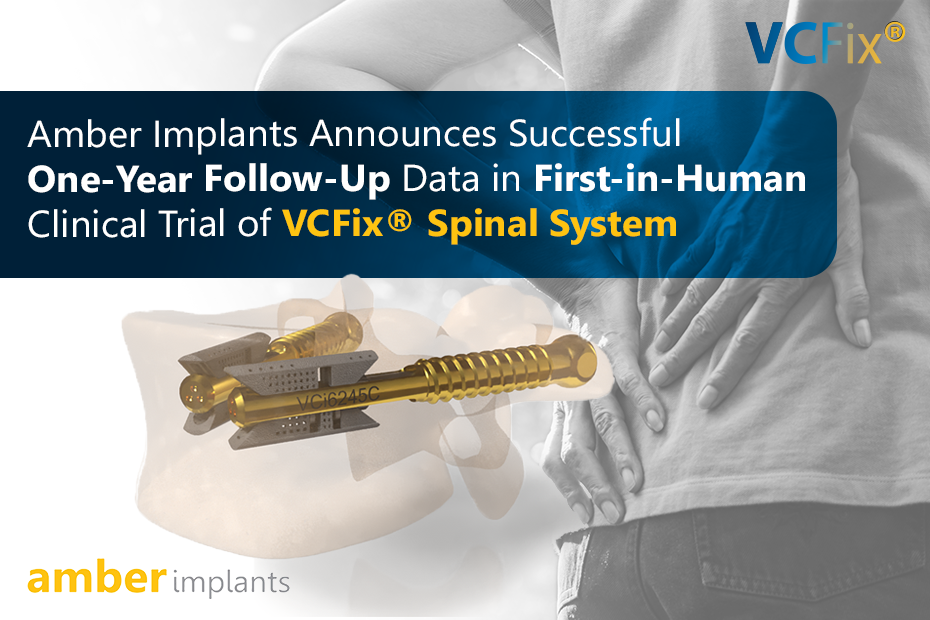
Successful completion and follow-up of the Fist-in-Human clinical trials. For more information, see the news item.
Finishing the pre-clinical activities and regulatory phase.
Receiving the series A fundraising.
Official kick off of the VCFix® project, including feasibility studies and receiving breakthrough designation
Our company was involved with more than 4 European level and national level researches and collaborations with 20 research partners.
Launching the company and recieving seed fund raising.

Introduction of VCFix® Spinal System with cement in the US market.



